Banruo Xianyu, Zeyu Lu, Huaping Xu*, Tianyu Li*
Biomaterials, 2026: p. 124099.
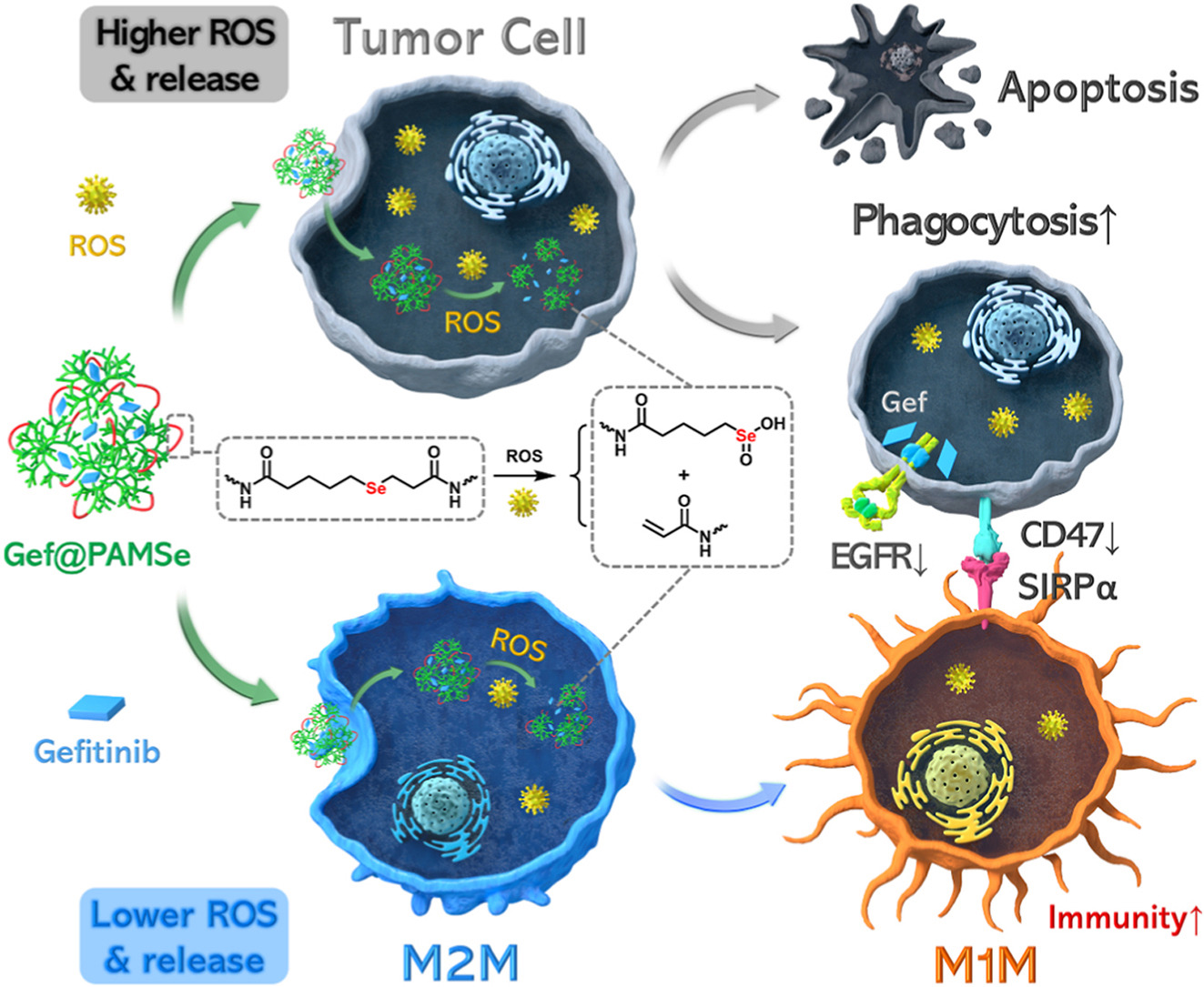
Tumor-associated macrophages (TAMs) have emerged as a promising immunotherapeutic target in non-small cell lung cancer (NSCLC). However, the indiscriminate cytotoxicity of chemotherapies and the immunosuppressive tumor microenvironment paradoxically impede this potential. To overcome these limitations, we engineered β-selenoester–crosslinked nanocapsules delivering Gefitinib, designed to induce opposite cell fates in cancer cells and macrophages. In cancer cells, the Se-C bond in β-selenoester is ultrasensitive under the intrinsic reactive oxygen species (ROS) level. It generates acrylates through selenoxide elimination reaction, which further depletes intracellular GSH to regenerate cytotoxic ROS. The ROS-triggered positive-feedback induces nanocapsule disassembly, enabling rapid Gefitinib release and apoptosis induction. The released Gefitinib also disrupts the CD47-SIRPα “don’t eat me” axis to enhance macrophage phagocytic activity. In macrophages, low ROS level limits Gefitinib exposure, but the selenium metabolites generated from the elimination reaction are sufficient to promote macrophage activation. This selective cell fate programming yielded no macrophage toxicity at cancer-cell IC50 levels and a 91.1 % tumor suppression in vivo. Collectively, this work demonstrates a divergent cell fate induction strategy based on β-selenoester–crosslinking for integrated TAM-mediated immunotherapy.